 For main group elements (i.e s-block and p-block elements), the valence electrons are the electrons present in the outermost orbit. Points to remember: Valence electrons are the electrons present in the outermost shell of an atom. By going through the periodic table, we see that the electron-dot symbol of atoms will never have more than eight dots around the atomic symbol. Periodic table with valence electrons is shown in the above image. With the next element, sodium, the process starts over with a single electron because sodium has a single electron in its highest-numbered shell, the n = 3 shell. Point to Remember: Mostly for transition and inner transition elements, the valence electrons are the electrons present in the shells outside the noble gas core.\] Even though the valence electrons in Group 3A elements are found in both s and p orbitals, it turns out that an elements row still corresponds to the energy level of that elements valence electrons. (See the above or below periodic table to see the valence electrons of inner transition metals). Identify elements that will have the most similar properties to a given element. Inner transition elements can have valence electrons ranging from 3 to 16. Explain the relationship between the chemical behavior of families in the periodic table and their valence electrons. In some inner transition metals, the electrons of incomplete d-orbitals are also considered as valence electrons. Visualize trends, 3D orbitals, isotopes, and mix compounds. It goes much deeper into the concept and delivers an extensive view of concepts like valence, charges, element names, etc. Hence, for inner transition elements, the electrons of both f-subshells as well as s-subshell behave like valence electrons. This video explains how to use the Periodic Table of Elements to determine the number of valence electrons in main group elements. Interactive periodic table showing names, electrons, and oxidation states. Our periodic table printable PDF does not contain the names of the elements only. The inner transition elements have incomplete f- subshells and they are very close to the outer s-subshell. (See the above or below periodic table to see the valence electrons of transition metals).Īlso the two bottom rows at the bottom of the periodic table are the inner transition elements (or f-block elements) also have the similar case. Hence, the transition elements (i.e d-block elements from group 3 to 12) can have more valence electrons ranging from 3 to 12. So, the electrons of both d-subshell and s-subshell behave like valence electrons. It is more difficult to find the valence electrons of transition elements as they have incompletely filled d-subshell and this d-subshell is very close to the outer s-subshell. What about valence electrons of transition and inner transition elements?įor the transition elements and inner transition elements, the case is more complicated. Well, this suits perfectly for the main group elements (i.e group 1, 2 and group 13 to 18), but what about the transition and inner transition elements? Label each column on the periodic table of elements from 1 to 18. The periodic table gives an atoms electron configuration based upon its location within the table. We can see this general size trend in the following periodic table. Electronegativity decreases as we move down a column in the periodic table. Hence, magnesium has 2 valence electrons. Moving down a column in the periodic table, valence electrons are held less tightly because they get further from the nucleus. Because much of the chemistry of an element is influenced by valence electrons, we would expect that these elements would have similar chemistry-and they do. A valence electron is an outer shell electron that can participate in a chemical bond with another atom. Valence is the number of connections an atom tends to form. They all have a similar electron configuration in their valence shells: a single s electron. 
Here, you can see that the highest principal quantum number is 3, and the total electrons in this principal quantum number is 2. Scientists soon observed patterns in the valence of the different elements. Hydrogen, at the upper left of the table, has an atomic number of 1. 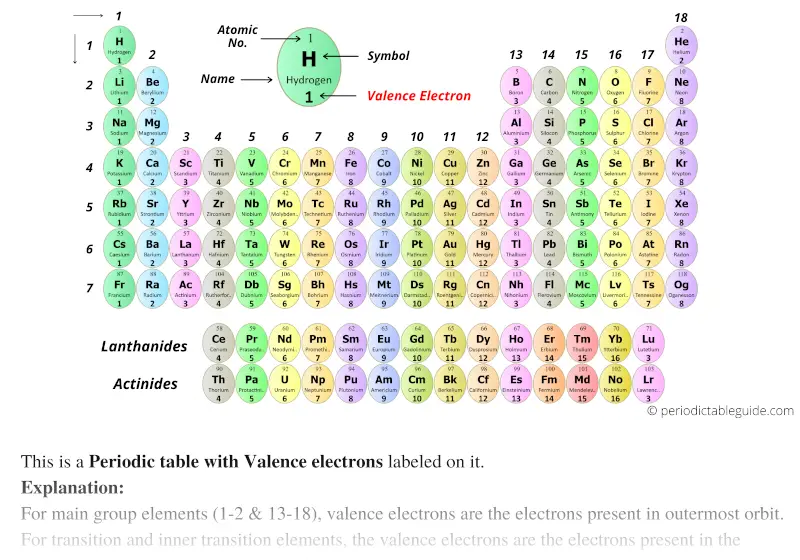
In this table, an elements atomic number is indicated above the elemental symbol. The electron configuration of magnesium is 1s 2 2s 2 2p 6 3s 2. The periodic table (see figure below) displays all of the known elements and is arranged in order of increasing atomic number. Valence electrons can also be determined as the electrons present in the shell with highest principal quantum number (n). Now I’ll show you the complete list of elements with electrons per shell. The magnesium element has 2 electrons in outermost orbit. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
